 
After improving his vacuum in the tube he set up two aluminum plates that, when connected to a battery, formed an electric field. This time however he set out to prove that the cathode ray carried an electronic charge. This effect would have looked similar to this:įor his second experiment, Thomson set his cathode ray up in a similar way as before in his first experiment. He later concluded that this proved that the ray and its negative electronic charge in inseparable. This showed the Thomson that the ray had moved with the magnet and was effected by its magnetic field. When he applied a magnet to the ray the electrometers read nothing. To do this, Thomson set up his cathode ray with electrometers on the other side of a metal cylinder, which he had cut a slit in. In Thomson's first experiment, he was determined to find out wether or not it was possible to separate the ray itself and the magnetic charge of the particles in the ray. Once attaching these wires to metal discs, and connecting them to a high voltage he pumped the air inside the glass tube to a very low pressure. He did this by getting a glass tube and inserting wires into two of the sides of the tube. He made a model almost identical to the one in the image above. The device is called an oscilloscope which is frequently used in medical treatment.The cathode ray, even though not invented by the physicist J. Used to measure changes in electrical voltage with time. They helped the transition from the industrial age to the digital.ģ. Vacuum tubes were initially used in the place of silicon transistors in electronics. In old displays, vacuum tubes were used by directing the beam of electrons using deflection plates, then the beam causes fluorescence on the screen which we see as white.Ģ. 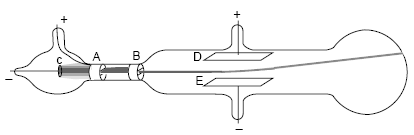
So, the cathode ray experiment is also commonly known as J.J. The above modified experiments were performed by J.J. This also helped scientists in finding the charge of electrons. Hence, energy of electron at point A = energy of electron at point B Applying a Magnetic FieldĬathode rays also get deflected from their path if a magnetic field is applied. At point B the electron stops due to the activation of stopping potential, so we apply the law of conservation of energy between the two points. When electrons move from one point to another, say from A to B. When we apply electric field in parallel but in the opposite direction to the cathode rays and if it is sufficiently high for the cathode rays to stop, then the magnitude of the applied voltage is called stopping potential. The magnitude of deviation is proportional to the magnitude of the electric field. When cathode rays hit from the cathode travel towards the anode and hit the anode, a fluorescence or glow is produced.Ĭathode ray deviates from its path due to the application of an electric field. When a high voltage is applied between the two electrodes of an evacuated discharge tube and the back of anode of the discharge tube is coated with a material, like zinc sulfide. They were later named electronsafter particles postulated by George Johnstone Stoney. Thomson measured the weight of cathode rays and showed that they were actually a beam of particles. 
German scientists Eilhard Wiedemann, Heinrich Hertz and Goldstein said they were some new form of electromagnetic radiation. Scientists Crooks and Arthur Schuster said they were electrically charged atoms. Scientists came up with two theories regarding cathode rays when they were originally discovered. Eugene Goldstein was the one who actually gave cathode rays their name. Cathode rays were first identified by a German physicist named Johann Hittorf when he realized that something was travelling through the tube. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
